PolyPid to Present Data from SHIELD II Phase 3 Trial Demonstrating Reduced Severity of Surgical Site Infection at Surgical Infection Society 2026 Annual Meeting
Analysis demonstrates statistically significant reduction in the severity of surgical wound infections, with potential to lower hospital resource utilization following abdominal colorectal surgery
PETACH TIKVA,
The presentation will be delivered by Dr.
The ASEPSIS score is a validated, objective clinical scoring system used to assess the severity of surgical wound infections. It integrates parameters such as wound appearance, antibiotic use, drainage of pus, isolation of pathogenic bacteria, and length of hospital stay. As previously reported in the SHIELD II topline results, treatment with D-PLEX100 together with the standard of care resulted in a 64% relative risk reduction (p=0.0103) in the proportion of patients with an ASEPSIS score greater than 20, the threshold indicative of clinically significant wound infection, compared to the standard of care alone.
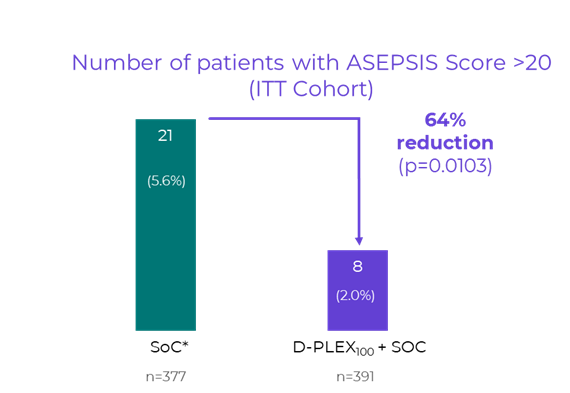
"The ASEPSIS score outcomes demonstrate not only that D-PLEX₁₀₀ reduces surgical site infections (“SSI”) events by 60%, but also that those infections that ultimately breakthrough are less severe and less complicated," said Dikla Czaczkes Akselbrad, Chief Executive Officer of
| Title: | Evaluation of ASEPSIS Score as a Secondary Endpoint in the Phase 3 SHIELD II Trial of D-PLEX100 in Colorectal Surgery |
| Presenter: | |
| Session: | Scientific Session III |
| Date/Time: | |
| Location: |
About D-PLEX100
D-PLEX100, PolyPid’s lead product candidate, is designed to provide local prolonged and controlled anti-bacterial activity directly at the surgical site to prevent surgical site infections (“SSIs”). Following the administration of D-PLEX100 into the surgical site, PolyPid’s delivery technology, Kynatrix, pairs with Active Pharmaceutical Ingredients, enabling a prolonged and continuous release of the broad-spectrum antibiotic doxycycline, resulting in a high local concentration of the drug for a period of 30 days for the prevention of SSIs, with additional potential to prevent SSIs caused by antibiotic-resistant bacteria at the surgical site. D-PLEX100 recently demonstrated positive results in the Phase 3 SHIELD II trial, achieving a statistically significant 60% (p=0.0013) relative risk reduction in SSI incidence following abdominal colorectal surgery with large incisions. D-PLEX100 received Breakthrough Therapy designation from the
About
Forward-looking Statements
This press release contains “forward-looking statements” within the meaning of the Private Securities Litigation Reform Act and other securities laws. Words such as “expects,” “anticipates,” “intends,” “plans,” “believes,” “seeks,” “estimates” and similar expressions or variations of such words are intended to identify forward-looking statements. For example, the Company is using forward-looking statements when it discusses the potential clinical and economic benefits of D-PLEX100, including its potential to reduce the incidence of severe wound infections, decrease wound complications and the use of intravenous antibiotics, enable earlier hospital discharge, and lower hospital resource utilization, and the potential role of D-PLEX100 in improving the prevention of SSIs. Forward-looking statements are not historical facts, and are based upon management’s current expectations, beliefs and projections, many of which, by their nature, are inherently uncertain. Such expectations, beliefs and projections are expressed in good faith. However, there can be no assurance that management’s expectations, beliefs and projections will be achieved, and actual results may differ materially from what is expressed in or indicated by the forward-looking statements. Forward-looking statements are subject to risks and uncertainties that could cause actual performance or results to differ materially from those expressed in the forward-looking statements. For a more detailed description of the risks and uncertainties affecting the Company, reference is made to the Company’s reports filed from time to time with the Securities and Exchange Commission, including, but not limited to, the risks detailed in the Company’s Annual Report on Form 20-F filed on
Company Contact:
908-858-5995
IR@Polypid.com
Investor Relations Contact:
Arx Investor Relations
North American Equities Desk
polypid@arxhq.com
A photo accompanying this announcement is available at https://www.globenewswire.com/NewsRoom/AttachmentNg/43a11cdb-f86b-4d84-bfb0-1870a0a2cb4e

Source: PolyPid Ltd.

